 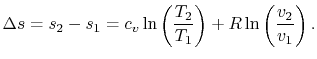
At the next level of approximation we correct the overestimation through inclusion of displacement covariances. 
At a first level of approximation, treating individual atomic displacements independently, our method may be applied using Debye–Waller B-factors, allowing diffraction experiments to obtain an upper bound on the thermodynamic entropy. We calculate the information-theoretic entropy from the expectation value of lnρ N. Ab initio molecular dynamics (AIMD) simulations generate ensembles of atomic configurations at finite temperature from which we obtain the N-body distribution of atomic displacements, ρ N. The vibrational entropy of a solid at finite temperature is investigated from the perspective of information theory. In conclusion, this advancement has the potential to reduce the evaluation the free energy to a simple modification to any procedure that evaluates the energy and the pair correlation function. We argue that a functional of the type, which is strictly applicable to pair potentials, is also suitable for first principles calculation of free energies from Born–Oppenheimer molecular dynamics performed at a single temperature. The pair more » functional approach gives the absolute entropy and free energy based on simulation output at a single temperature without thermodynamic integration. Approximate excess entropy functionals are developed and compared to results from thermodynamic integration. We apply the Kirkwood approximation, which is commonly used for fluids, to both fluids and solids. The stationarity and concavity of the excess entropy functional are discussed and related to the Gibbs–Bugoliubov inequality and to the free energy. This result extends Henderson’s theorem, which states that the free energy is a temperature dependent functional of the density and pair correlation. We prove that, within the class of pair potential Hamiltonians, the excess entropy is a universal, temperature-independent functional of the density and pair correlation function. We form the compound experimentally and confirm that it develops as a single BCC phase from the melt, but that it transforms reversibly at lower temperatures. Our first-principles calculation of free energy predicts that the configurational entropy stabilizes a single body-centered cubic (BCC) phase from more » T = 1700 K up to melting, while precipitation of a complex intermetallic is favored at lower temperatures. We demonstrate phase stability in the multicomponent alloy system of Cr–Mo–Nb–V, for which some of its binary subsystems are subject to phase separation and complex intermetallic-phase formation. Some HEAs exhibit desirable properties, such as high specific strength, ductility, and corrosion resistance, while challenging the scientist to make confident predictions in the face of multiple competing phases. 
High entropy alloys (HEAs) are multicomponent compounds whose high configurational entropy allows them to solidify into a single phase, with a simple crystal lattice structure. In conclusion, first-principles more » density functional theory (DFT) calculations on the electronic and magnetic structures reveal that the anti-ferromagnetism of Mn atoms in CoFeMnNi is suppressed especially in the CoFeMnNiAl HEA because Al changes the Fermi level and itinerant electron-spin coupling that lead to ferromagnetism. This phase transition leads to the significant enhancement of the saturation magnetization (M s), e.g., the CoFeMnNiAl alloy has M s of 147.86 Am 2/kg. The dramatic change in phase structures from face-centered-cubic (FCC) to ordered body-centered-cubic (BCC) phases, caused by adding Al, Ga, and Sn in CoFeMnNiX alloys, originates from the potent short-range chemical order in the liquid state predicted by ab initio molecular dynamics (AIMD) simulations. Based on the high-entropy effect, a system of CoFeMnNiX (X = Al, Cr, Ga, and Sn) magnetic alloys are designed and investigated. 
Magnetic materials with excellent performances are desired for functional applications. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
